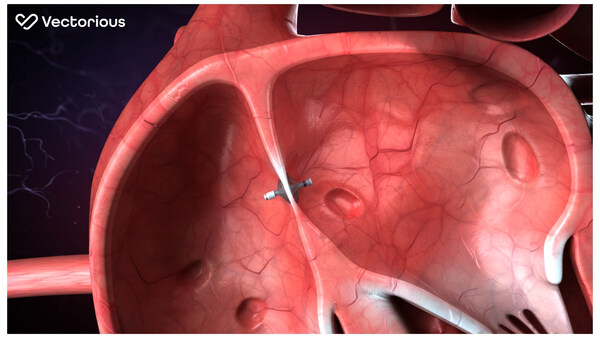
Femasys Inc. (NASDAQ: FEMY), a biomedical company focused on meeting the significant unmet needs for women worldwide with a broad portfolio of in-office, accessible solutions, including a lead, late-clinical stage product candidate and innovative therapeutic and diagnostic products, today highlighted that its FemaSeed intratubal insemination product, a revolutionary approach to enhancing fertilization, is expected to serve as a first-line therapeutic option that is substantially lower cost with significantly less risk than assisted reproductive methods, such as in vitro fertilization (IVF) or intracytoplasmic sperm injection (ICSI). FemaSeed has been designed to deliver sperm reliably and safely, directly into the fallopian tube where conception occurs.
“Femasys remains committed to providing accessible and affordable options and the timing of FemaSeed’s launch is particularly relevant given the uncertainty following the Alabama Supreme Court ruling on February 16th. Following our substantial cash infusion in the fourth quarter 2023, we are now funded into the second half of 2025 and on track to make FemaSeed, available later this year to women and their doctors, as a first-line treatment option that does not involve handling of embryos,” said Femasys’ CEO Kathy Lee-Sepsick.